SN2 Reactions
Relevant Definitions
Nucleophile - a chemical species that donates a pair of electrons to an electrophile to form a chemical bond during a reaction. Negatively charged molecule.
Electrophile - a reagent in a chemical reaction that accepts an electron pair in order to bond to a nucleophile. Positively charged molecule.
Bimolecular Reaction - involved the collision of two molecules and the rate of the reaction depends on the product of both species involved.
Inversion of Configuration - inversion of a chiral center in a molecule during a chemical reaction.
Steric Hindrance - the size of the other molecules attached to the central carbon hindering the nucleophile from reaching the central carbon.
Nucleophile - a chemical species that donates a pair of electrons to an electrophile to form a chemical bond during a reaction. Negatively charged molecule.
Electrophile - a reagent in a chemical reaction that accepts an electron pair in order to bond to a nucleophile. Positively charged molecule.
Bimolecular Reaction - involved the collision of two molecules and the rate of the reaction depends on the product of both species involved.
Inversion of Configuration - inversion of a chiral center in a molecule during a chemical reaction.
Steric Hindrance - the size of the other molecules attached to the central carbon hindering the nucleophile from reaching the central carbon.
Example of SN2 Reaction with (R)-2-Bromobutane in Water
Water is a highly soluble compound and an excellent nucleophile.
Steps involved in Reaction Mechanism: 1 Step
Step 1:
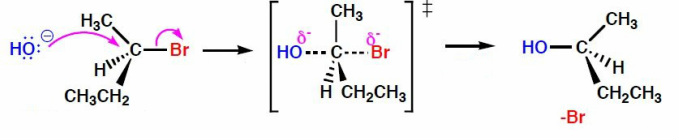
During an SN2 reaction, there is only 1 step in which the nucleophile attaches to the central carbon and the bond between the leaving group and central carbon breaks. Hydroxide, which in this case acts as the nucleophile attaches to the central carbon on the opposite side of the leaving group.
Steps involved in Reaction Mechanism: 1 Step
Step 1:
During an SN2 reaction, there is only 1 step in which the nucleophile attaches to the central carbon and the bond between the leaving group and central carbon breaks. Hydroxide, which in this case acts as the nucleophile attaches to the central carbon on the opposite side of the leaving group.
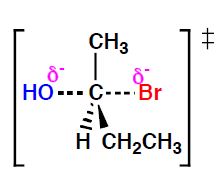
The image above shows the "Transition State". In this state the bond between the oxygen and carbon is partially formed and the bond between the bromine and carbon is partially broken. This is when the carbon atom begins to invert.
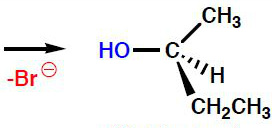
The bond between the oxygen and carbon is now formed and the bromine ion has departed. The configuration of the carbon is now inverted.
Reaction Limitations
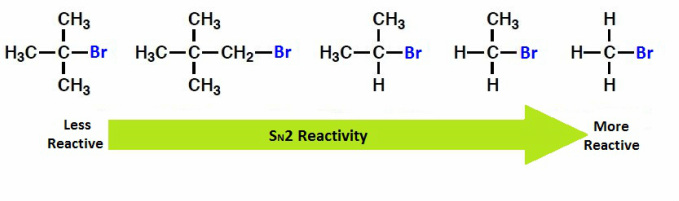
Steric hindrance makes a compound less reactive because bulky substituents make it difficult for a nucleophile to attach to a carbon from the opposite side of the leaving group.
Steric hindrance makes a compound less reactive because bulky substituents make it difficult for a nucleophile to attach to a carbon from the opposite side of the leaving group.
SN2 reactions are favored when strong nucleophiles are present. These are compounds that are strongly electronegative and either have a lone pair of electrons to donate or are negatively charged ions.
The best leaving groups weak bases. As you go down a column on a periodic table, the more likely it is to leave and is thus a better leaving group. The weaker the base the faster the reaction.